Shotty lymph nodes after infection4/23/2024  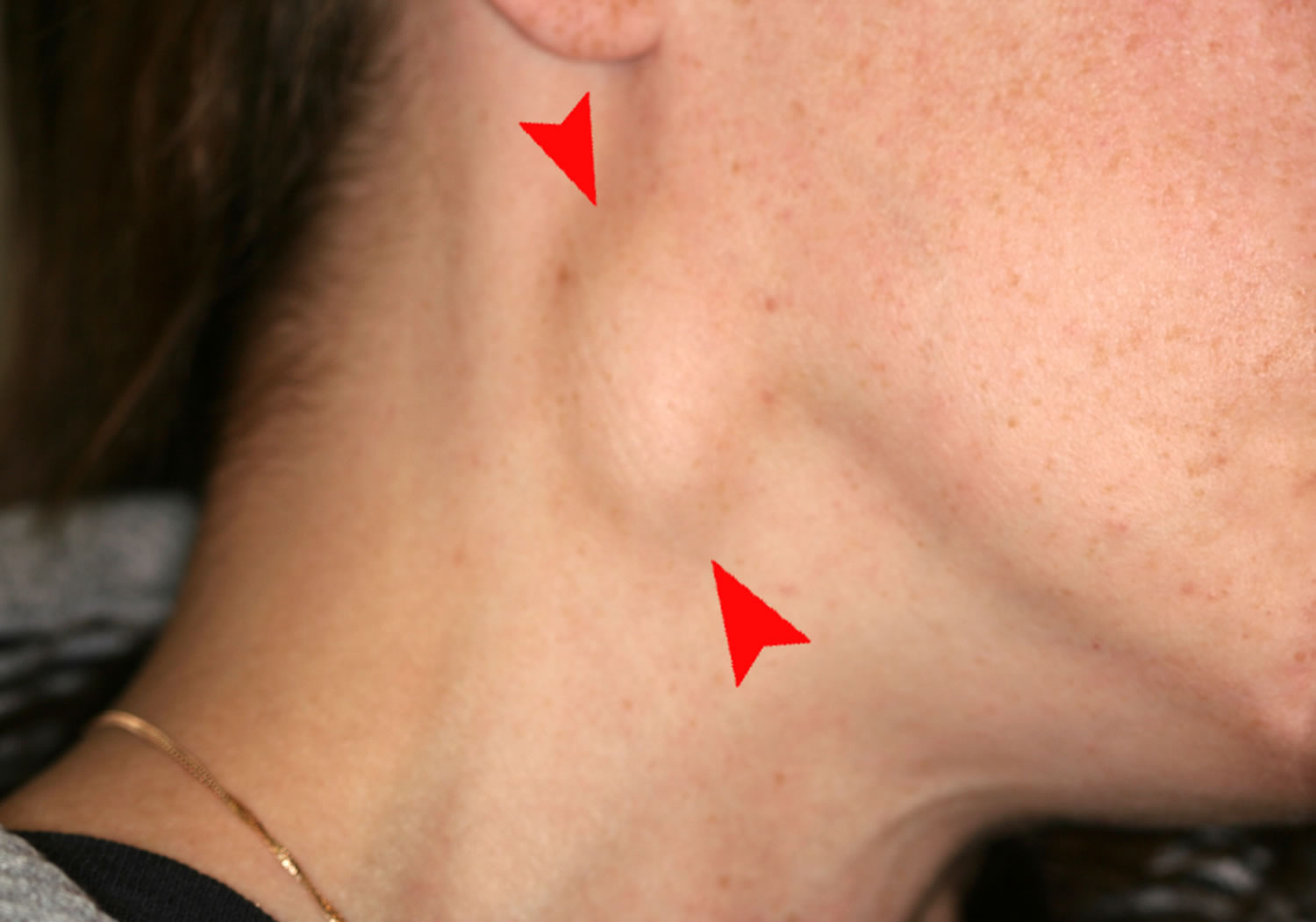
These cellular gaps also serve to draw in interstitial fluid (red-green gradient filled arrows in ( B, D– F)), including any bacteria present, by increasing intraluminal volume and thus decreasing pressure. 
Hydrostatic pressure forces fluid out of capillaries at the arterial end (indicated by red-green gradient filled arrows in ( A, C, E, F)) which swells the interstitium and causes the tethered lymphatics to open wide gaps between endothelial cells that can accommodate bacteria. However, ( C) bacteria are much too large to passively enter vascular capillaries in most tissues and instead ( D) pass into the more permeable initial lymphatics. Though small molecules (≤10 nm diameter) are preferentially absorbed from the interstitial space into the blood capillaries rather than lymphatic capillaries, uptake into the lymphatics rises with increasing molecular size. By contrast, ( B, D) the initial lymphatics have an incomplete basement membrane, lack smooth muscle, and have buttonlike wide gap junctions between endothelial cells, as well as anchoring filaments that are tethered to surrounding tissue. In tissue, fluid leaks from ( A, C) vascular capillaries and venules are characterised by tight junctions between endothelial cells, a complete basement membrane, presence of pericytes, and occasional smooth muscle fibres. Many of these bacteria accumulate in the spleen and liver, and these organs play important roles in the clearance of bacteraemia.įluid exchange and bacterial uptake in the vasculature. Bacteria that have entered the bloodstream can now seed any tissue or other organs in the body. A number of bacteria leave the lymph node through the efferent lymphatic vessel and drain with efferent lymph into collecting ducts before entering the bloodstream via the subclavian veins. Although bacteria are initially present in lower numbers than in local draining lymph nodes, virulent strains can rapidly replicate within the lymph node niche, despite the activity of resident and recruited phagocytes and other leucocytes. Bacteria transit in efferent lymphatics to reach distant sequential draining lymph nodes and again accumulate in the sinuses. However, numerous bacteria escape the filtering activity of the lymph node and exit through the efferent lymphatic. Bacteria accumulate in the subcapsular sinus and, particularly, the medullary sinuses of the lymph node. Bacteria transit extracellularly along the lymphatics into collecting lymphatics and then enter local draining lymph nodes in afferent lymph. 
Bacteria inoculated into tissue (here streptococcal intramuscular infection of a mouse is depicted, but similar results are obtained using other infection routes and models) preferentially and passively enter the initial lymphatics (green channels) rather than blood vessels (dark red and blue channels). Path of bacterial dissemination following intramuscular inoculation of bacteria. We discuss the anatomy of the lymphatics and other factors that influence bacterial dissemination, as well as the consequences of underappreciated bacterial lymphatic metastasis on disease and immunity.īacteraemia bacteria bacterial dissemination immunity infection invasion lymph nodes lymphatic metastasis lymphatic system lymphatics. Here, we summarise the routes and mechanisms by which an increasing variety of bacteria are acknowledged to transit through the lymphatic system, including those that do not necessarily require internalisation by host cells. Accordingly, it is timely to reconsider the role of lymph nodes as absolute barriers to bacterial dissemination in the lymphatics. Recently, we have demonstrated that extracellular bacterial lymphatic metastasis of virulent strains of Streptococcus pyogenes drives systemic infection. There is little direct data on live virulent bacteria, instead understanding is largely inferred from studies investigating immune responses to viruses or antigens in lymph nodes. Despite offering an obvious conduit for pathogen spread, the lymphatic system has long been regarded to bar the onward progression of most bacteria. Involvement of the lymphatics in the pathogenesis of bacterial infections is not well studied. Lymphatic vessels permeate tissues around the body, returning fluid from interstitial spaces back to the blood after passage through the lymph nodes, which are important sites for adaptive responses to all types of pathogens. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
